by staff | Feb 27, 2018 | Addiction, Drug Policy, News, Opioids
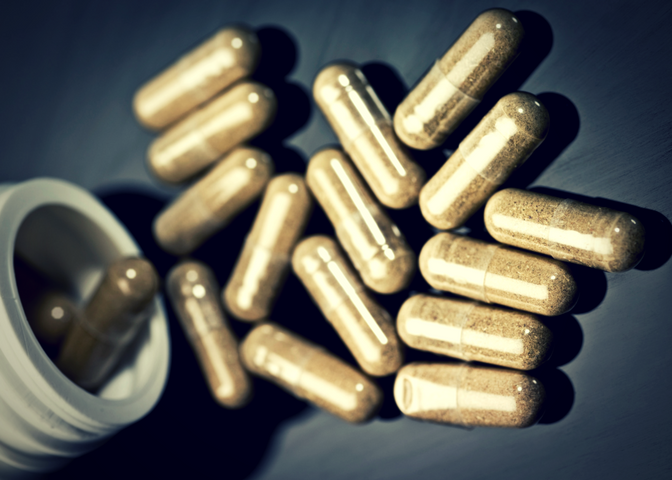
This past week the Center for Disease Control and Prevention (CDC) issued a statement warning people to stay away from Kratom while health officials investigate the possible connection between the plant and a nationwide outbreak of salmonella.
Multi-State Salmonella Outbreak
So far 11 people have been hospitalized as a result of this recent salmonella outbreak, and the CDC believes that kratom may actually be responsible for the recent chain of illnesses.
Since October, 28 cases of salmonella were recorded in 20 states, including:
- California
- Florida
- New York
- Ohio
- Massachusetts
- Michigan
- Alabama
- Arizona
- Colorado
- Louisiana
- Oklahoma
- Oregon
- North Dakota
- North Carolina
- South Carolina
- Kansas
- Kentucky
- Utah
- Tennessee
- Pennsylvania
So far there have been no deaths, but nearly a dozen people have ended up needing to be hospitalized.
Most people infected with salmonella develop symptoms with 12 to 72 hours of exposure to the bacteria. Some symptoms include:
- Diarrhea
- Fever
- Abdominal cramps
According to the advisory released by the CDC, 11 people affected by the salmonella outbreak were interviewed. Out of those 11 people, 8 of them admit to consuming kratom. That is a 73% connection so far. These individuals had taken the plant via:
Therefore, at this time kratom is the primary suspect in the CDC’s investigation. The advisory states:
“Epidemiologic evidence indicates that kratom is a likely source of this multistate outbreak. At this time, CDC recommends that people not consume kratom in any form because it could be contaminated with salmonella.”
But it wasn’t just the CDC. The very next day the Food and Drug Administration (FDA) issued a statement announcing the recall of “kratom-containing dietary supplements” manufactured and distributed by Divinity Products Distribution of Grain Valley, Missouri. This manufacturer is not yet determined to be the cause of the outbreak, but the company voluntarily recalled its kratom products and promised to stop selling them.
CDC, FDA, and DEA vs Kratom
At this time the FDA is encouraging other kratom companies to follow the Missouri company’s lead. They urge other manufacturers to- “take swift action to remove these products from circulation to protect the public.” The FDA Commissioner Scott Gottlieb states,
“To protect the public health, we’ll continue to affirm the risks associated with kratom, warn consumers against its use and take aggressive enforcement action against kratom-containing products.”
Proponents of kratom do argue that the FDA has been working especially hard in the last few years to prove that kratom is a threat to public health. In early February the FDA claimed it found evidence that certain compounds in kratom interact with the body’s opioid receptors. This led the agency to conclude that “compounds in kratom make it so it isn’t just a plant—it’s an opioid.” In America, the use of this plant is actually already banned in 6 states. The DEA also considers it a drug of concern. Even though back in October of 2016 the DEA announced they would not be banning kratom and giving it a schedule 1 label.
However, the American Kratom Association and other kratom advocates are willing to keep pushing back against the FDA. Many kratom users claim it is useful for helping addicts self-medicate to lessen the withdrawals when getting off heroin and other opioids. Yet, there is not enough research out there yet to fully endorse this claim, and a lot more would have to be done to legitimize it.
Either way, because kratom products are very loosely regulated by the FDA, it isn’t hard to understand why they are asking people to stop using the plant until they have been able to identify the source of the bacteria.
So far, no specific brands or suppliers have been singled out, but health officials are still urging people to be safe and avoid kratom products.
Many may still debate the benefits and the risks with kratom. Either way, both sides agree that more research is needed. Whether or not the drug should be banned is still a question that has yet to be answered. The one thing we do know is that kratom is a mind-altering substance, so using it might be detrimental to some people trying to recover from addiction. Right now, it doesn’t seem safe for anyone, anyway. If you or someone you love is struggling with substance abuse or addiction, please call toll-free now. We want to help.
CALL NOW 1-888-922-5398
by Justin Mckibben | Nov 2, 2017 | Maintenance Drugs, Methadone, Naloxone, News, Prescription Drugs, Suboxone, Vivitrol

While the nation is still struggling to find the right strategy to climb out of the opioid crisis in U.S. it seems many are holding onto the idea that Big Pharma is going to save us from the destruction they helped create. While we can agree that evidence-based medical assistance in recovery is a useful tool, some seem to think that the only fix for a pill problem is more pills, or in this case, more needles.
Just recently, after President Trump declared the opioid crisis a “National Health Emergency”, the advisory committee to the U.S. Food and Drug Administration (FDA) voted 18-1 that a new injectable drug called RBP-6000 could benefit addicts and the lower of two doses studied had an “acceptable” safety profile.
So what does this mean for the opioid epidemic efforts? Is Indivior a miracle injection that is going to make the epidemic more manageable, or is it another kind of Methadone or Suboxone that is just keeping people hooked?
What is RBP-6000?
For a little background, RBP-6000 is an experimental drug designed to help fight America’s growing opioid addiction crisis. It is described as a sustained-release buprenorphine. It is designed to be delivered once a month as a subcutaneous injection. The compound solidified once in contact with bodily fluids and releases buprenorphine over time.
If approved, it will be the first monthly injectable buprenorphine treatment. When creating the drug the manufacturers studied two dosing regimens.
In one, 300 milligrams were given once a month for six months.
In the other, two doses of 300 milligrams were followed by four doses of 100 milligrams.
According to reports, there was only a minute difference in effectiveness between the two doses, and they drug company acknowledge that the higher dose of RBP-6000 caused more side effects. Looking closely into some of the reports from the research, some side-effects include:
-
Headache
-
Constipation
-
Nausea
-
Injection site pruritus
-
Vomiting
-
Insomnia
-
Upper respiratory tract infection
While these side effects were not noted in an extremely high percentage of those tested, they are still relevant to consider until there is more extensive data available. So far, the report says the safety profile is consistent with that of Suboxone.
So RBP-6000 is like Suboxone using the delivery of Vivitrol; another injectable drug used to try and block the effects of opioids.
Big Pharma Making Big Moves
This is one sign of how Big Pharma is stepping in to make a buck off of the opioid epidemic yet again.
Indivior is the company behind RBP-6000. It was originally part of Reckitt Benckiser as the Buprenorphine division but has since split off to be a specialty pharmaceutical company. Indivior already sells Suboxone Film, a product which combines buprenorphine and naloxone. Suboxone is a maintenance drug widely used in America to try and curb the effects of opioid addiction and withdrawal, but Suboxone is known to have its own side-effects and withdrawals. Some even attest that Suboxone is itself addictive and very difficult to get off of.
Two months ago the share prices for Indivior took a deep hit after a U.S. court ruling clearing the way for a generic rival to Suboxone Film. So, with new competition on the way in the Suboxone market, Indivior put a renewed focus on another maintenance drug to bring to market.
Some analysts already expect that RBP-6000 could capture around 30% of the broader buprenorphine market. Some believe this new form of injectable buprenorphine could generate annual sales of around $700 million by 2021.
Jefferies sees potential sales of $1.3 billion by 2025.
So now the push for this new drug is boosting its sales prospects as competitors threaten revenues from Suboxone sales.
To learn more about why Suboxone isn’t the easier answer some people think it is, download our FREE E-BOOK
“5 Things No One Tells You About Suboxone”
DOWNLOAD FREE E-BOOK
Waiting for FDA Final Ruling
This endorsement on behalf of RBP-6000 comes less than a week after FDA Commissioner Scott Gottlieb announced the agency’s plans to promote extensive use among opioid addicts of less harmful opioids such as methadone and buprenorphine, the active ingredient in RBP-6000.
However, some of the FDA panelists still would like to see more data about which patients should be given the higher dose. Other panelists say they would like to have it available, even though they lack of data supporting any additional benefit of higher doses. So even though the drug is shown to have side-effects in higher doses, and there is limited information on how these side-effects impact the individual, the FDA is considering to let this drug pass the grade.
The FDA’s decision is set to be made by November 30th, although typically the agency follows the recommendations of its advisory panels. So it may very well already be a done deal.
Why It Matters
Again, with respect to the importance of offering alternatives to particularly dangerous opioids like heroin or fentanyl. It is important to have resources for the purpose of preserving of life. Keeping people alive long enough to get help is crucial. But we also have to see that this is not a miracle cure.
Methadone, Suboxone, and Vivitrol may serve a purpose for some in a certain capacity. However, these drugs are not an adequate substitute for comprehensive addiction treatment. Each one of these methods should be accompanied by a holistic treatment plan that addresses all aspect of addiction, including emotional and mental health.
Drugs like RBP-6000 may help subdue the more serious symptoms from the physical side of addiction, but they can also present their own risks.
It is important to offer safe medical assistance, but we have to remember that these Big Pharma companies are not selling us instant cures to opioid addiction. This isn’t even the first time we have seen a new drug come out to try and treat the opioid problem from companies that are closely related or directly responsible for narcotic medications that helped fuel the issue.
Remember, there is not a quick fix for this problem. We cannot prescribe our way out of the problem. It is going to take better treatment resources beyond more medications.
Drugs like RBP-6000 from Indivior are not necessarily a bad thing, but they also may not be the kind of treatment people should focus on. Instead, there are safe medical treatment options that offer holistic resources and mental health services. A lasting recovery begins with effective treatment. If you or someone you love is struggling, please call toll-free now.
CALL NOW 1-888-922-5398
by Justin Mckibben | Jul 11, 2017 | Drug Abuse, Opana, Opioids, Prescription Drugs

Oxymorphone is a powerful semi-synthetic opioid analgesic developed in Germany back in 1914. Since the painkiller’s introduction into the medical world it has gone by many brand names, such as:
-
Numorphan (suppository and injectable solution)
-
Opana ER (extended-release tablet)
-
Opana IR (immediate-release tablet)
-
O-Morphon in Bangladesh by Ziska pharmaceutical ltd
In the midst of the opioid epidemic in America many pharmaceutical drugs are now under close scrutiny for their addictive potential. Now, a public announcement has come to light explaining that in June of 2017, the U.S. Food and Drug Administration asked the manufacturer of Opana ER, a Oxymorphone medication, to remove the product from the market.
What is Opana?
Opana or Oxymorphone is meant for use as:
- Moderate to severe pain relief
- Preoperative medication to alleviate apprehension
- Medication to maintain anaesthesia
- As a obstetric analgesic
Oxymorphone extended-release tablets are designated for managing chronic pain. These tablets are also only for people already on a regular schedule of strong opioids for an extended time.
Oxymorphone immediate-release tablets are recommended for breakthrough pain for people on the extended-release version.
Endo International PLC is the Big Pharma empire based in Dublin, Ireland that manufactures Opana.
- 2006- Opana approved for use in the United States
- 2012- Endo changed the drug’s formulation to try to make it harder to abuse. The FDA approved sales of the new version, however the FDA prohibited Endo International from marketing Opana as abuse deterrent.
- 2013- the Centers for Disease Control and Prevention (CDC) reported an illness associated with intravenous abuse of oral Opana ER in Tennessee.
- 2015- Reports in Austin, Indiana indicated an outbreak of HIV was caused by recreational injection of Opana
Endo primarily makes generic medicines, as well as a number of brand-name specialty drugs. Endo reported that Opana ER last year posted net sales of $159 million.
Opana ER Abuse
The FDA has taken a revolutionary stance after deciding that the risks greatly outweighed the benefits of Opana use.
In a statement on Thursday Endo International PLC said it will voluntarily stop selling the pills. However, the drug-maker does not necessarily agree with the conclusion made by the FDA, adding in their statement that the extended-release opioid is safe and effective when used as intended, and that Endo still believes Opana ER’s benefits outweigh its risks.
And yet, FDA advisers are firm after reviewing the safety of Opana ER and voting 18-8 against keeping it on the market.
The agency said it had perceived a “significant shift” from individuals abusing the drug recreationally by crushing and snorting the pill to injecting it instead. Besides the stories in Indiana and Tennessee, Opana was also called out for contributing to the rising rates of drug overdose and overdose deaths.
According to the FDA there were no generic versions of the reformulated Opana ER on the market, as of June. However, there are two generics of earlier versions of Opana on sale, called Oxymorphone.
The Big News For Big Pharma
The big news here is that this is the first time the U.S. Food and Drug Administration has actively taken steps to remove a currently marketed opioid pain medication from sale due to the drugs abuse and the related public health consequences!
Some see this as a major move in the fight toward overcoming the opioid addiction problem gripping the nation.
Thus, Endo International PLC has committed to working with the FDA to try to minimize disruption for patients relying on the drug for pain relief. Of course those prescribed to the drug for medical reasons will also need to be provided with alternative treatments.
But the FDA isn’t done yet. The U.S. Food and Drug Administration claims that its agencies will be reviewing other opioid painkillers and could take further action to regulate or even eliminate dangerous opioid medications like Opana ER.
Taking steps to reduce the impact of this epidemic is a step in the right direction. Big Pharma might be in for a bit of a shakedown from the FDA as they attempt to reduce the amount of dependence on opioids. If you or someone you love is struggling, please call toll-free now.
CALL NOW 1-888-922-5398




